 The oxygen has a formal charge of 6 - 4 - 2 = 0 (same ordering of terms). The central carbon has a formal charge of 4 (valence electrons) - 0 (lone pairs) - 4 (bonds) = 0. All atoms in the molecule have zero formal charge, the "happiest" situation for any molecule. 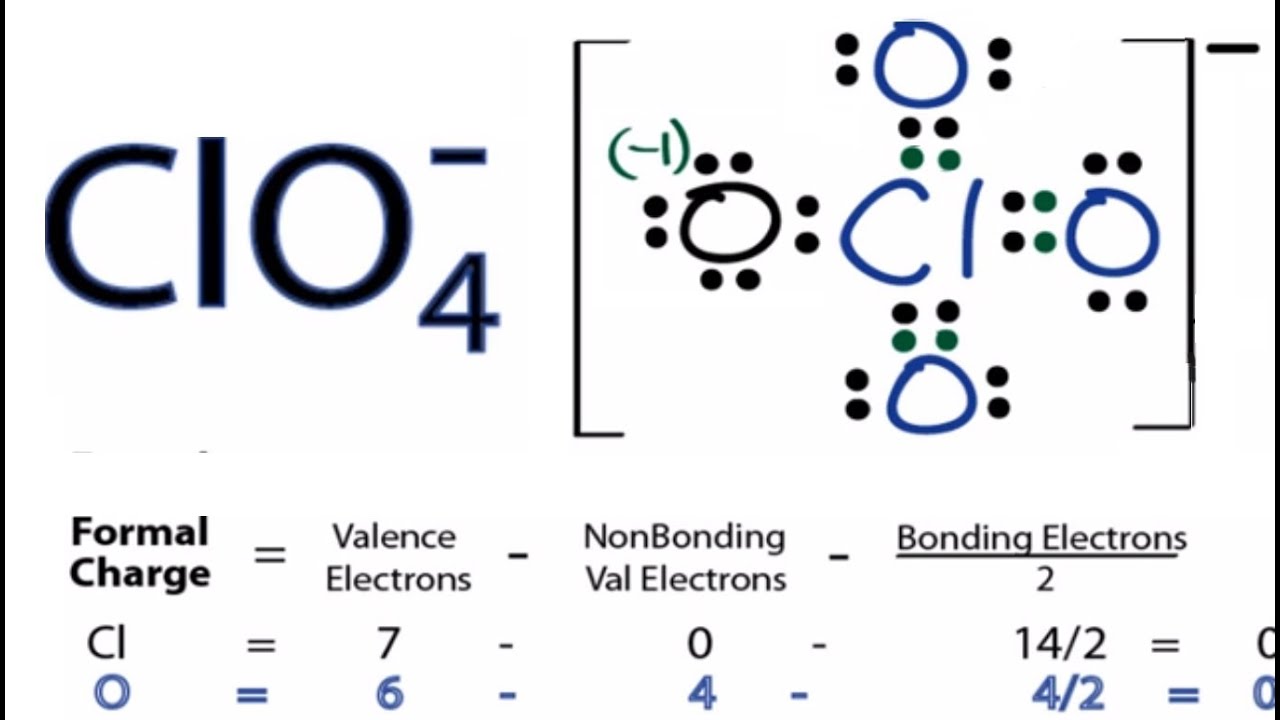
The sum of the formal charges of all atoms. Each hydrogen atom has one electron as a neutral atom, no lone pairs and shares one bond, for a formal charge of zero. We can double-check formal charge calculations by determining the sum of the formal charges for the whole structure. It has no lone pairs, and it shares four bonds, so the formal charge is zero. The formal charge on an atom in a molecule or ion is equal to the total number of valence electrons in the free atom minus the total number of electrons of lone pairs (non-bonding electrons) minus half of the total number of shared electrons bonding electrons. The carbon in CH 4 has four electrons as a neutral atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
